Advertisements
Advertisements
Question
Describe the shapes of BF3 and BH4–. Assign the hybridisation of boron in these species.
Solution
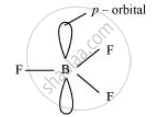
BF3
As a result of its small size and high electronegativity, boron tends to form monomeric covalent halides. These halides have a planar triangular geometry. This triangular shape is formed by the overlap of three sp2 hybridised orbitals of boron with the sporbitals of three halogen atoms. Boron is sp2 hybridised in BF3.
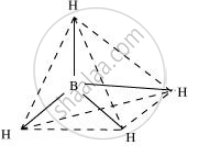
BH4–
Boron-hydride ion (BH4–) is formed by the sp3 hybridisation of boron orbitals. Therefore, it is tetrahedral in structure
APPEARS IN
RELATED QUESTIONS
Why does boron trifluoride behave as a Lewis acid?
Consider the compound, CCl4. How will CCl4 behave with water? Justify.
What do you understand by Allotropy?
What do you understand by catenation?
Write a balanced equation for BF3 + LiH → ?
Consider the compound, BCl3. How will BCl3 behave with water? Justify.
Which of the following statements is INCORRECT?
