Advertisements
Advertisements
Question
Why does boron trifluoride behave as a Lewis acid?
Solution
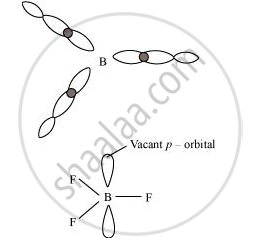
The electric configuration of boron is ns2 np1. It has three electrons in its valence shell. Thus, it can form only three covalent bonds. This means that there are only six electrons around boron and its octet remains incomplete. When one atom of boron combines with three fluorine atoms, its octet remains incomplete. Hence, boron trifluoride remains electron-deficient and acts as a Lewis acid.
APPEARS IN
RELATED QUESTIONS
Consider the compound, CCl4. How will CCl4 behave with water? Justify.
Describe the shapes of BF3 and BH4–. Assign the hybridisation of boron in these species.
What do you understand by Allotropy?
What do you understand by catenation?
Write a balanced equation for BF3 + LiH → ?
Consider the compound, BCl3. How will BCl3 behave with water? Justify.
Which of the following statements is INCORRECT?
