Advertisements
Advertisements
Question
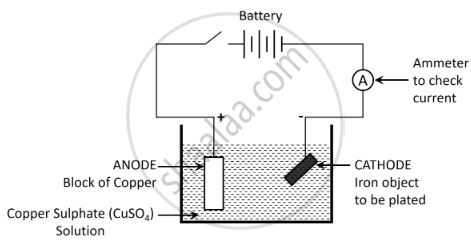
Draw a labelled diagram to show how iron is electroplated with copper.
Solution
APPEARS IN
RELATED QUESTIONS
Name the following :
The process of coating of iron with zinc
Give reasons why In the electroplating of an article with silver, the electrolyte sodium argentocyanide solution is preferred over silver nitrate solution.
State the electrode reaction at the anode during electrolysis of:
Aqueous copper sulphate solution.
How is impure copper purified by electrolysis ? Explain.
Element X is a metal with valency 2. Element Y is a non-metal with valency 3.
(a) Write equations to show how X and Y form ions.
(b) If Y is diatomic gas, write the equation for the direct combination of X and Y to form a compound.
(c) Write two applications of electrolysis in which anode diminish in mass.
(d) If the compound formed between X and Y is melted and an electric current passed through the molten compound, the element X will be obtained at the _____ and the Y at the ________of the electrolytic cell. (Provide the missing words).
A solution of silver nitrate is a good electrolyte but is not used for electroplating an article with silver.
Copy and complete the following table :
| Anode | Electrolyte | |
| Purification of copper |
Describe what happens to the iron object and the copper rod.
Write two applications of electrolysis in which the anode diminishes in mass.
What ions must be present in a solution used for electroplating a particular metal?
