Advertisements
Advertisements
Question
Draw the structure of α-D glucopyranose.
Solution

RELATED QUESTIONS
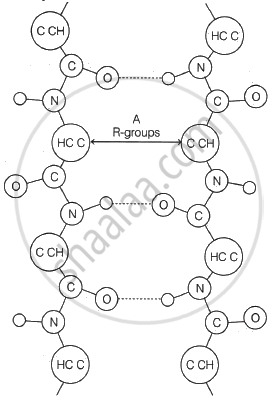
Draw a neat diagram for the following:
Haworth formula of glucopyranose.
Glucose on oxidation with dilute nitric acid gives _______________
What is the action of the following reagents on glucose?
hydroxylamine
Draw the Haworth projection structure of the following.
α-D-(–)-Fructofuranose
Which of the following type of carbohydrates cannot be hydrolyzed?
Which of the following carbohydrate is sweet to taste?
One mole of a carbohydrate on hydrolysis gives one mole of glucose and one mole of galactose. Identify the carbohydrate.
Stachyose is ____________.
\[\ce{CH3 - CH2 - CH2 - Br ->[Na][dry ether] A ->[Cl2] B}\]
Identify the product B in the above reaction.
Which one of the following is Tetrose sugar?
Which following reagent is used to detect presence of five hydroxyl groups in a glucose molecule?
Which one of the following sugar does NOT have same empirical fonnula as that of carbohydrate?
What is the number of hydroxyl groups present in lactic acid?
Which among the following reagents is used to confirm the presence of carbonyl group in glucose?
Which among the following sugars does not reduce Tollen's reagent?
What is the quantity of glucose obtained when 68.4 g of sucrose is hydrolyzed in laboratory under ideal condition?
(Molar mass of sucrose = 342 g mol−1)
How many moles of acetic acid are obtained in the reaction when one mole glucose is treated with excess acetic anhydride?
Which among the following is a product of hydrolysis of one mole raffinose?
Lactose is made of ______.
A molecule of stachyose contains how many carbon atoms?
The two monosaccharides in a disaccharide are held together by ______ bonds.
Corn is immersed in boiling water. It is then cooled, and the solution becomes sweet. It is due to ______.
Formation of gluconic acid from glucose by oxidation using Br2 water.
The glycosidic linkage present in maltose is ______.
Why carbohydrates are generally optically active.
Why are carbohydrates generally optically active?
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
