Advertisements
Advertisements
Question
During thunderstorm, rain water contains nitric acid. Explain with reactions.
Solution
During a lightning discharge or thunderstorm, the nitrogen present in the atmosphere reacts with oxygen to form nitric oxide.
\[\ce{N2 + O2 <=> 2NO}\]
Nitric oxide is further oxidized to nitrogen dioxide.
\[\ce{2NO + O2 <=> 2NO2}\]
The nitrogen dioxide dissolves in atmospheric moisture in the presence of oxygen in the air and forms nitric acid, which is washed down by the rain and combines with the salt present on the surface of the earth.
\[\ce{4NO2 + 2H2O + O2 -> 4HNO3}\]
That is why during thunderstorms, rainwater contains nitric acid.
APPEARS IN
RELATED QUESTIONS
Fill in the blanks using the appropriate words given below :
(Sulphur dioxide, nitrogen dioxide, Nitric oxide, Sulphuric acid)
Hot, concentrated nitric acid reacts with sulphur to form ______________.
Write balanced equations for action of warm water on magnesium nitride
What is aqua fortis?
What is fixation of Nitrogen?
Write the balanced equation for the following:
Action of heat on AgNO3
Write down the word equation or balanced equation for the action of concentrated nitric acid on copper.
Write the equation of the following reaction.
Sulphur can be converted to sulphuric acid using concentrated nitric acid.
Write equation to show the reaction between the following:
Copper oxide and dilute nitric acid.
Fill in the blank using the appropriate words given below:
Cold, dilute nitric acid reacts with copper to give ______.
The diagram given below is a representation of the Industrial preparation of Nitric acid by Ostwald’s process. With respect to the process answer the following questions:
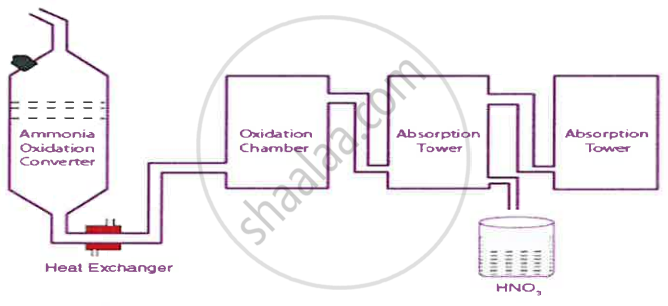
- Write the temperature and the catalyst required during the catalytic oxidation of ammonia.
- Give balanced chemical equation for the reaction occurring duringthe conversion of nitrogen dioxide to nitric acid.
