Advertisements
Advertisements
Question
Explain an example of a Polar Covalent bond.
Solution
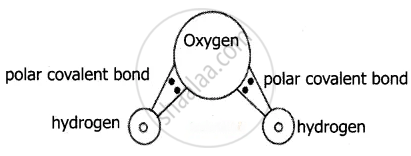
In water (H2O) molecule, covalent bond exists between Hydrogen and Oxygen. Being more electronegative Oxygen carries negative charge and Hydrogen carries positive charge. In a water molecule Oxygen carries a negative charge (anionic in nature) and hydrogen’s carries positive charge (cationic in nature) which forms a polar covalent bond. The size of an oxygen atom is comparatively higher than a hydrogen atom, hence it polarizes the molecule towards itself i.e., it attracts shared pair of bonding electrons towards itself and makes the bond to be more polarized. Hydrogen which is comparatively a small-sized cation makes the bond to be polarized better.
APPEARS IN
RELATED QUESTIONS
In the molecule OA = C = OB, the formal charge on OA, C and OB are respectively.
Draw the Lewis structure for the following species.
\[\ce{SO^{2-}4}\]
Draw the Lewis structure for the following species.
HNO3
Explain the bond formation if BeCl2 and MgCl2.
What is the Polar Covalent bond?
Draw the Lewis structure for the following species.
NO3–
Draw the Lewis structure for the following species.
\[\ce{NO^-3}\]
Draw the Lewis structure for the following species.
\[\ce{NO^-3}\]
Draw the Lewis structures for the following species.
\[\ce{NO^-3}\]
Draw the Lewis structure for the following species.
\[\ce{SO^2-_4}\]
