Advertisements
Advertisements
Question
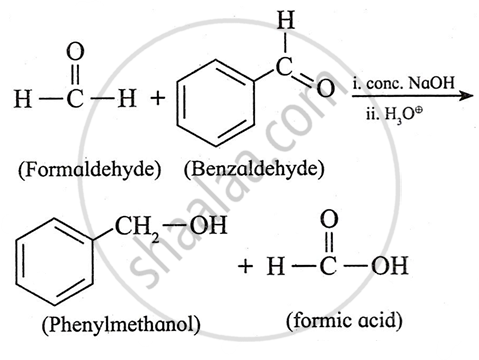
Explain Cross Cannizzaro reaction.
Chemical Equations/Structures
Explain
Solution
When a mixture of formaldehyde and non-enolisable aldehyde (aldehyde with no α-hydrogen) is treated with a strong base, fomaldehyde is oxidized to formic acid while the other non-enolisable is reduced to alcohol. Formic acid forms sodium formate with NaOH. On acidification, sodium formate is converted into formic acid.
For example
shaalaa.com
Is there an error in this question or solution?
