Advertisements
Advertisements
Question
Explain quantitatively the order of magnitude difference between the diamagnetic susceptibility of N2 (~5 × 10–9) (at STP) and Cu (~10–5).
Advertisements
Solution
Magnetic susceptibility: It is the property of the substance which shows how easily a substance can be magnetised. It can also be defined as the ratio of intensity of magnetisation (I) in a substance to the magnetic intensity (H) applied to the substance, i.e., XM = I/H.
According to the problem, we have
Density of nitrogen `ρ_(N_2) = (28 g)/(22.4 L) = (28 g)/(22400 "cc")`
Also. density of copper `ρ_(C_u) = (8 g)/(22.4 L) = (8 g)/(22400 "cc")`
So, ratio of both densities
`(ρ_(N_2))/(ρ_(Cu)) = 28/22400 xx 1/8 = 16 xx 10^-4`
Also given `(chi_(N_2))/(chi_(Cu)) = (5 xx 10^-9)/10^-5 = 5 xx 10^-4`
`(chi_m)_("Fero") ≈ 10^3`
and `(chi_m)_("Para") ≈ 10^-5`
⇒ `((chi_m)_("Fero"))/((chi_m)_("Para")) = 10^3/10^-5 = 10^8`
APPEARS IN
RELATED QUESTIONS
Out of the two magnetic materials, 'A' has relative permeability slightly greater than unity while 'B' has less than unity. Identify the nature of the materials 'A' and 'B'. Will their susceptibilities be positive or negative?
A meter gauge train is heading north with speed 54 km/hr in earth's magnetic field 3 x 10-4T. The e.m.f. induced across the axle joining the wheels is ..........
(a) 0.45 mV
(b) 4.5 mV
(c) 45 mV
(d) 450 m V
The susceptibility of a magnetic material is 0·9853. Identify the type of magnetic material. Draw the modification of the field pattern on keeping a piece of this material in a uniform magnetic field.
Which of the following substances are diamagnetic?
Bi, Al, Na, Cu, Ca and Ni
The susceptibility of a magnetic material is − 2.6 × 10−5. Identify the type of magnetic material and state its two properties.
The relative magnetic permeability of a magnetic material is 800. Identify the nature of magnetic material and state its two properties.
A domain in a ferromagnetic substance is in the form of a cube of side 1 µm. If it contains 8 × 1010 atoms and each atomic dipole has a dipole moment of 9 × 10-24 Am2 then the magnetisation of the domain is ______.
For a paramagnetic substance, the magnetic susceptibility is ______.
Magnetic material can be easily magnetized if magnetic susceptibility is ______.
The given figure represents a material which is ______.
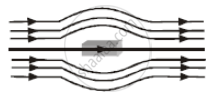
Metals getting magnetised by orientation of atomic magnetic moments in external magnetic field are called ______.
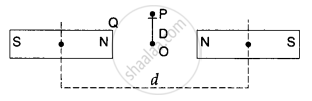
Two identical bar magnets are fixed with their centres at a distance d apart. A stationary charge Q is placed at P in between the gap of the two magnets at a distance D from the centre O as shown in the figure. The force on the charge Q is ______.
If a charged particle at rest experience no electromagnetic force
- Assertion (A): Diamagnetic substances exhibit magnetism.
- Reason (R): Diamagnetic materials do not have a permanent magnetic dipole moment.
Which of the following cannot modify an external magnetic field as shown in the figure?

Explain the origin of paramagnetism on the basis of atomic structure.
Explain the Domain theory in brief.
Magnetic susceptibility of a diamagnetic substance ______.
