Advertisements
Advertisements
Question
Explain the terms Dialysis
Solution
Dialysis - The process of removing a dissolved substance from a colloidal solution by the means of diffusion through a membrane is known as dialysis. This process is based on the principle that ions and small molecules can pass through animal membranes unlike colloidal particles.
APPEARS IN
RELATED QUESTIONS
What modification can you suggest in the Hardy-Schulze law?
Explain what is observed An electrolyte, NaCl is added to hydrated ferric oxide sol.
Explain what is observed Electric current is passed through a colloidal sol?
Explain the term Electrophoresis
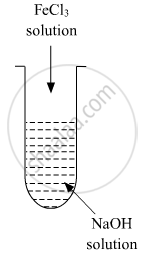
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?
What happens when a lyophilic sol is added to a lyophobic sol?
(i) Lyophobic sol is protected.
(ii) Lyophilic sol is protected.
(iii) Film of lyophilic sol is formed over lyophobic sol.
(iv) Film of lyophobic sol is formed over lyophilic sol.
What causes brownian motion in colloidal dispersion?
Assertion: Colloidal solutions show colligative properties.
Reason: Colloidal particles are large in size.
The important factor in the stabilization of a colloidal solution is ______
The right option for the statement “Tyndall effect is exhibited by”, is ______.
Define coagulation.
Define the following term:
Coagulation
Why a negatively charged sol is obtained when AgNO3 solution is added to KI solution?
Which of the following is a correct statement?
Coagulation value of the electrolytes AlCl3 and NaCl for As2S3 sol are 0.093 and 52 respectively. The No. of times AlCl3 has greater coagulating power than NaCl is ______.
6.84 g Al2(SO4)3 is needed to coagulate 2.5 L of As2S3 sol completely in 2.0 hrs. The coagulation value of Al2 (SO4)3 is ______.
Lyophilic sols are more stable than lyophobic sols because ______.
