Advertisements
Advertisements
Question
Explain what is observed An electrolyte, NaCl is added to hydrated ferric oxide sol.
Solution
When NaCl is added to ferric oxide sol, it dissociates to give Na+ and Cl- ions. Particles of ferric oxide sol are positively charged. Thus, they get coagulated in the presence of negatively charged Cl- ions.
APPEARS IN
RELATED QUESTIONS
Define the following terms: Associated colloids
Out of MgCl2 and AlCl3, which one is more effective in causing coagulation of negatively charged sol and why?
Out of BaCl2 and KCl, which one is more effective in causing coagulation of a negatively charged colloidal Sol? Give reason.
Explain what is observed Electric current is passed through a colloidal sol?
Explain the terms Dialysis
Explain the terms Tyndall effect.
Answer the following question.
Why are medicines more effective in the colloidal state?
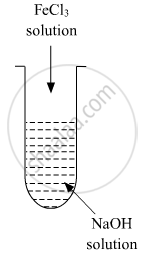
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?
Which of the following will show Tyndall effect?
The values of colligative properties of colloidal solution are of small order in comparison to those shown by true solutions of same concentration because of colloidal particles ______.
What happens when a lyophilic sol is added to a lyophobic sol?
(i) Lyophobic sol is protected.
(ii) Lyophilic sol is protected.
(iii) Film of lyophilic sol is formed over lyophobic sol.
(iv) Film of lyophobic sol is formed over lyophilic sol.
Why do we add alum to purify water?
What causes brownian motion in colloidal dispersion?
Tyndall effect is observed in
Which of the following is used for removing change on colloidal solution?
Gold number is associated with:-
Define coagulation.
Why a negatively charged sol is obtained when AgNO3 solution is added to KI solution?
Which of the following is a correct statement?
