Advertisements
Advertisements
Question
Explain, why is water used as a coolent in motor car radiators?
Solution
Water has the highest specific heat capacity of 4.2 Jg-1 °C-1 and its temperature does not rise beyond 100°C. Thus, it can absorb large amount of heat from the working engine, which is then radiated out through radiator Thus, on low temperature the engine works efficiently.
APPEARS IN
RELATED QUESTIONS
How can a temperature in degree Celsius be converted into S.I. unit of temperature?
Describe an experiment to show that there is absorption of heat energy when the ice melts.
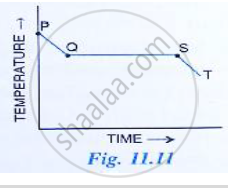
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
Give scientific reasons for the following:
It is much easier to skate on rough ice than on glass.
40 g of ice at -16°C is dropped into water at 0°C, when 4 g of water freezes into ice. If specific heat capacity of ice is 2100 J/kg°C, what will be the latent heat of fusion of ice?
Why does the heat supplied to a substance during its change of state not cause any rise in its temperature?
Find the final temperature when a mass of 80g of water at 100°C is mixed with a mass of 40g of water at 25°C.
Conduction is the way of heat transfer which takes place in a ______.
