Advertisements
Advertisements
Question
Functional group present in terylene polymer is ______.
Solution
Functional group present in terylene polymer is ester.
APPEARS IN
RELATED QUESTIONS
Answer the following in one sentence.
Name some chain-growth polymers.
Answer the following in one sentence.
Define the term: Synthetic fibres
Answer the following in one sentence.
Give one example each of copolymer and homopolymer.
Answer the following.
Write the names of classes of polymers formed according to intermolecular forces and describe briefly their structural characteristics.
Attempt the following:
Classify the following polymer as straight-chain, branched-chain and cross-linked polymers.
\[\begin{array}{cc}\ce{- (CH2 - CH -)_\text{n}}\\
\phantom{.....}|\\\ce{\phantom{.......}CN}\end{array}\]
Answer the following.
Explain in detail free radical mechanism involved during preparation of addition polymer.
Addition polymer among the following is ___________
Homopolymer among the following is _____________
Write the number of carbon atoms present in the monomer used for preparation of nylon 6 polymer.
Explain the term copolymers with examples.
Explain the classification of polymers on the basis of origin.
Identify the monomers used in the preparation of glyptal.
The total number of electrons around the carbon atom of methyl tree radical are ______.
Which of the following is a natural polymer?
Cuprammonium rayon and viscose rayon are ____________ polymers.
Identify semisynthetic polymer.
Which of the following is INCORRECT regarding terylene?
Which among the following polymers belongs to the class elastomers?
Which of the following properties is of thermoplastic polymer?
Identify thermoplastic polymer from the following.
The polymer used in making synthetic hair wigs is made up of ______.
Lactic acid and glycollic acid are the monomers used for the preparation of the polymer.
Write the structure of monomer used in the preparation of
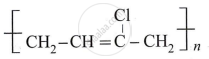
Write the classification of polymers on the basis of intermolecular forces.
Write names of two natural polymers.
