Advertisements
Advertisements
Question
Fused alumina is reduced to aluminium by electrolytic reduction, since ______.
Options
Alumina is highly stable
Alumina is least stable
Alumina is not reduced by drying agents.
Alumina is not reduced by reducing agents.
Solution
Fused alumina is reduced to aluminium by electrolytic reduction, since Alumina is highly stable.
APPEARS IN
RELATED QUESTIONS
For sodium hydroxide, explain its significance in the extraction of aluminium.
Name the following:
The materials used as electrodes in the electrolytic extraction of aluminium.
Give reason for the following:
Aluminium oxide cannot be reduced by carbon.
For the substance listed below, explain its role in the extraction of aluminium: Graphite
The following question relate to the extraction of aluminium by electrolysis.
Explain why is it necessary to renew the anode periodically.
Describe the role played in the extraction of aluminum : Graphite
Answer the following question:
Write the chemical formula of one main ore of iron and aluminium.
What impurities are present in aluminium ore?
The following sketch illustrates the process of conversion of Alumina to Aluminium:
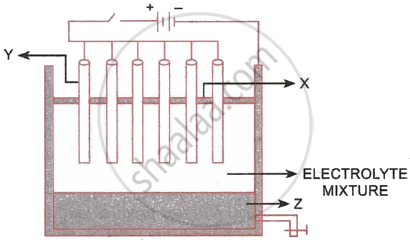
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
Given below in column A is a schematic diagram of the electrolytic reduction of alumina. Identify the parts labelled as A, B and C with the correct options from the Column B.
| column A | column B | |
 |
1. | Platinum |
| 2. | Anode | |
| 3. | Cathode | |
| 4. | Electrolyte mixture | |
| 5. | Bauxite |
