Advertisements
Advertisements
Question
Give balanced equations for the following reactions:
(i) Acetaldehyde is heated with hydroiodic acid in the presence of red phosphorous.
(ii) Calcium acetate is subjected to dry distillation.
(iii) Sodium ethoxide is treated with ethyl bromide.
(iv) Benzaldehyde is treated with sodium bisulphite.
Solution 1
(i)
\[\ce{\underset{\text{Acetaldehyde}}{CH3CHO} + 4HI ->[red P][\Delta]\underset{\text{Ethane}}{CH3CH3} + H2O + 2I2}\]
(ii)
\[\ce{\underset{\text{Calcium acetate}}{(CH3COO)2Ca} ->[\Delta]\underset{\text{Acetone}}{CH3COCH3} + CaCO3}\]
(iii)
\[\ce{\underset{\text{Sodium ethoxide}}{C2H5ONa} + \underset{\text{Ethylbromide}}{C2H5Br} -> \underset{\text{Diethylether}}{C2H5OC2H5} + NaBr}\]
(iv)
\[\begin{array}{cc}
\phantom{.................................}\ce{H}\\
\phantom{.................................}|\\
\ce{\underset{\text{Benzaldehyde}}{C6H5CHO} + \underset{\text{Sodium bisulphite}}{NaHSO3} -> C6H5-C-OH}\\
\phantom{................................}|\\
\phantom{.....................................}\ce{\underset{\text{Benzaldehyde sodium bisulphite}}{SO3Na}}
\end{array}\]
Solution 2
(i)
\[\ce{\underset{\text{Acetaldehyde}}{CH3CHO} + 4HI ->[red P][\Delta]\underset{\text{Ethane}}{CH3CH3} + H2O + 2I2}\]
(ii)
\[\ce{\underset{\text{Calcium acetate}}{(CH3COO)2Ca} ->[\Delta]\underset{\text{Acetone}}{CH3COCH3} + CaCO3}\]
(iii)
\[\ce{\underset{\text{Sodium ethoxide}}{C2H5ONa} + \underset{\text{Ethylbromide}}{C2H5Br} -> \underset{\text{Diethylether}}{C2H5OC2H5} + NaBr}\]
(iv)
\[\begin{array}{cc}
\phantom{.................................}\ce{H}\\
\phantom{.................................}|\\
\ce{\underset{\text{Benzaldehyde}}{C6H5CHO} + \underset{\text{Sodium bisulphite}}{NaHSO3} -> C6H5-C-OH}\\
\phantom{................................}|\\
\phantom{.....................................}\ce{\underset{\text{Benzaldehyde sodium bisulphite}}{SO3Na}}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Predict the main product of the following reactions: \[\ce{CHO + CH_3CHO->[dil NaOH]}\]
\[\ce{CHO + CH_3CHO->[dil NaOH]}\]
Write the main product formed when propanal reacts with the following reagents:
(ii) Dilute NaOH
`CH_3CH_2CHO + NaOH(dil)-> `
Give balanced chemical equations for the following reactions:
(1) Acetaldehyde reacts with hydrogen cyanide.
(2) Acetone reacts with phenylhydrazine.
(3) Acetic acid is treated with ethanol and a drop of conc. H2SO4.
Give one chemical test each to distinguish between the following pairs of compounds:
(1) Acetone and benzaldehyde.
(2) Phenol and benzoic acid.
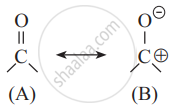
In the following resonating structures A and B, the number of unshared electrons in valence shell present on oxygen respectively are ______.
Choose the most correct option.
Aldol condensation is
Choose the most correct option.
Which one of the following has the lowest acidity?
Choose the most correct option.
Diborane reduces
Answer the following in one sentence.
What are aromatic ketones?
What are aliphatic aldehydes?
