Advertisements
Advertisements
Question
Give one chemical test each to distinguish between the following pairs of compounds:
(1) Acetone and benzaldehyde.
(2) Phenol and benzoic acid.
Solution
(1)
Benzaldehyde gives Tollen's reagent test. i.e., a silver mirror with Tollen's reagent but acetone does not.
(2)
Phenol gives a white precipitate with bromine water but benzoic acid does not.
APPEARS IN
RELATED QUESTIONS
What are ketones?
A compound used as pistachio flavour in ice cream is ___________
Answer the followiiig questions:
Identify the compounds A, B, C and D in the given reaction:
Predict the main product of the following reactions: \[\ce{CHO + CH_3CHO->[dil NaOH]}\]
\[\ce{CHO + CH_3CHO->[dil NaOH]}\]
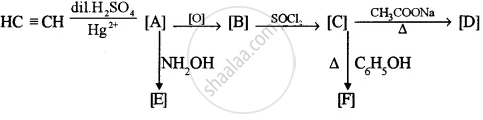
Identify the compounds A, B, C, D, E and F:
Give balanced equations for the following reactions:
(i) Acetaldehyde is heated with hydroiodic acid in the presence of red phosphorous.
(ii) Calcium acetate is subjected to dry distillation.
(iii) Sodium ethoxide is treated with ethyl bromide.
(iv) Benzaldehyde is treated with sodium bisulphite.
Choose the most correct option.
In the Wolff-Kishner reduction, alkyl aryl ketones are reduced to alkyl benzenes. During this change, ketones are first converted into ______.
Choose the most correct option.
Aldol condensation is
Answer the following in one sentence.
What are aromatic ketones?
Which of the following is the product of reaction of ethane nitrile with Grignard reagent followed by acid hydrolysis?
