Advertisements
Advertisements
Question
Give example for the following type of organic reaction.
β - elimination
Solution
In this reaction, two substituents are eliminated from the molecule, and a new C – C double bond is formed between the carbon atoms to which the eliminated atoms/groups are previously attached. Elimination reaction is always accompanied with a change in hybridisation.
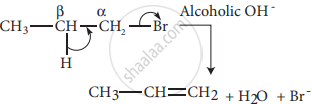
Example:
n – Propyl bromide on reaction with alcoholic KOH gives propene. In this reaction, hydrogen and Br are eliminated.
APPEARS IN
RELATED QUESTIONS
What is the hybridisation state of benzyl carbonium ion?
Which of the group has highest + I effect?
Assertion: Tertiary Carbocations are generally formed more easily than primary Carbocations ions.
Reason: Hyper conjucation as well as inductive effect due to additional alkyl group stabilize tertiary carbonium ions.
Which of the following represent a set of nucleophiles?
Which of the following species does not acts as a nucleophile?
The geometrical shape of carbocation is ______.
Write a short note on resonance.
Write a short note on hyperconjucation.
Show the heterolysis of a covalent bond by using curved arrow notation and complete the following equation. Identify the nucelophile.
CH3 – Br + KOH →
Explain the electromeric effect.
