Advertisements
Advertisements
Question
Give one example in the case where supplying energy [given below] is necessary for a chemical reaction.
Pressure
Solution
Pressure:
It causes the reacting molecules, to come together and collide with greater intensity.
Example:
Production of ammonia.
\[\ce{N2 + 3H2 ⇌[Fe - 450°C][200 atoms.] 2 NH3 + \triangle}\]
APPEARS IN
RELATED QUESTIONS
Translate the following statement into chemical equation and then balance it.
Hydrogen gas combines with nitrogen to form ammonia
Complete and balance the following equation:
CA (OH)2 + .............  CaCO3 + H2O
CaCO3 + H2O
Ammonia reacts with oxygen to form nitrogen and water. Write a balanced chemical equation for this reaction. Add the state symbols for all the reactants and products.
Balance the following chemical equation:
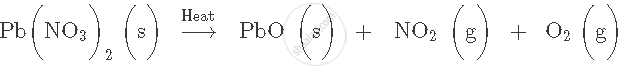
Write the balanced chemical equation of the following reaction. potassium bicarbonate + sulphuric acid → potassium sulphate + carbon dioxide + water
Write the balanced chemical equation of the following reaction.
sodium chloride + manganese dioxide + sulphuric acid → sodium hydrogen sulphate + manganese sulphate + water + chlorine.
A student mixes sodium sulphate powder in barium chloride powder. What change would the student observe on mixing the two powders? Justify your answer and explain how he can obtain the desired change.
Balance the following simple equation:
KHCO3 → K2CO3 + H2O + CO2
Write a balanced equation for the following word equation:
Potassium bicarbonate → Potassium carbonate + Water + Carbon dioxide
Give a balanced equation by the partial equation method, [steps are given below].
Oxidation of Lead [II] Sulphide by Ozone
- Ozone first decomposes to give molecular oxygen & nascent oxygen.
- Nascent oxygen then oxidises lead [II] sulphide to lead [II] sulphate.
