Advertisements
Advertisements
Question
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Solution
Oxidation of ethanol by K2Cr2O7:
\[\ce{CH3CH2OH->[K2Cr2O7|H^+][2{[O]}]\underset{\text{Ethanoic acid}}{CH3COOH} + H2O}\]
APPEARS IN
RELATED QUESTIONS
Give a balanced chemical equation for Preparation of ethane from Sodium propionate
Organic compounds having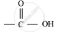 functional group are known as
functional group are known as
Write the name of the following functional group:

Generally _______ is called spirit.
An organic compound ‘A’ is widely used as a preservative and has the molecular formula C2H4O2. This compound reacts with ethanol to form a sweet-smelling compound ‘B’.
Name the process.
Ethanol reacts with sodium and forms two products. These are
A gas is evolved when ethanol reacts with sodium. Name the gas evolved and also write the balanced chemical equation of the reaction involved.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
