Advertisements
Advertisements
Question
Give the chemical formula of sodium hydroxide
Solution
It denotes in a compound, the number of atoms of element present.
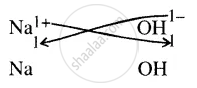
∴ Formula = \[\ce{NaOH}\]
APPEARS IN
RELATED QUESTIONS
What happens to the number of valence electrons in the atoms of elements as we go down in a group of the periodic table?
State its position in the periodic table?
What is the main characteristic of the last element in each period of the periodic table?
Name an alkali metal in period 3 and halogen in period 2.
Answer the following question.
Define the atomic size. Give its unit of measurement. In the modern periodic table what trend is observed in the atomic radius in a group and a period and why is it so?
An X element with atomic number 11 and Y element with atomic number 13 belongs to the third period in the modern periodic table. With this information, give the answers of the below questions.
- Which element has more metallic character from these two elements?
- What is the valency of each X and Y elements?
- Atom size of Y element is smaller than atom size of X element. Explain.
State whether true or false. If false, correct the statement.
Lanthanides and actinides are kept at the bottom of the periodic table because they resemble each other but they do not resemble with any other group elements.
An element which is an essential constituent of all organic compounds belongs to
Noble gases are placed in the ______ group in the modern periodic table.
A part of modern periodic table is given below. On its basis, answer the following questions:
| Group No. → | 1 | 2 | 13 | 14 | 15 | 16 | 17 | 18 |
| Period ↓ | ||||||||
| 2 | A | B | ||||||
| 3 | E | D | F | C |
Which of the element is a Metalloid?
