Advertisements
Advertisements
Question
\[\ce{H2O2 -> H2O + O2}\]
This represents ______.
Options
Oxidation of H2O2
Reduction of H2O2
Disproportionation of H2O2
Acidic nature of H2O2
MCQ
Fill in the Blanks
Solution
\[\ce{H2O2 -> H2O + O2}\]
This represents Disproportionation of H2O2.
Explanation:
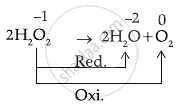
It is known as a disproportionation reaction because the same elements are oxidised and reduced.
shaalaa.com
Oxidation Number - Types of Redox Reactions
Is there an error in this question or solution?
