Advertisements
Advertisements
Question
How are metals refined by the electrolytic process? Describe the electrolytic refining of copper with the help of a neat labelled diagram.
Solution
The process of electrolysis is used in electrolytic refining. This is the most widely used method for the refining of impure metals. Several metals like copper, zinc, tin, lead, nickel, chromium, gold, silver are refined by this process.
Electrolytic refining of copper:
An electrolytic refining tank consists of an anode, a cathode and an electrolyte (acidified copper sulphate solution). A thick rod of an impure metal is made the anode and attached to the positive terminal. A thin rod of a pure metal is made the cathode and attached to the negative terminal. When electricity is passed through the cell, impure metal from the anode gets dissolved in the electrolyte. The metal ions from the electrolyte move to the cathode and get deposited there as pure metal. While the soluble impurities dissolve in the electrolyte, the insoluble ones deposit under the anode as anode mud.

When electricity is passed through the electrolytic cell, following reactions take place at the anode and cathode:
(i) Impure copper atoms from the anode lose two electrons to form copper ions and enter copper sulphate electrolyte.
Anode: 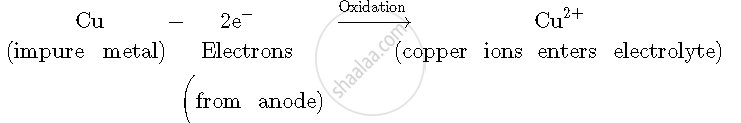
Copper ions from the copper sulphate electrolyte move to the cathode (negatively charged) by gaining two electrons from the cathode. Thus, they get reduced to copper atoms.
Cathode:

As the process continues, the cathode (pure metal collected) progressively becomes thicker and anode (impure metal) keeps getting thinner. The metal obtained is 99.99 percent pure.
APPEARS IN
RELATED QUESTIONS
Name one method for the refining of metals.
What is meant by refining of a metal? Name the most widely used method for the refining of impure metals obtained by various reduction processes. Describe this method with the help of a labelled diagram by taking the example of any metal.
Draw a neat labelled diagram.
Magnetic separation method.
Draw a neat labelled diagram.
Electrolytic reduction of alumina.
Which of the following process to be carried out to avoid the
formation of greenish layer on brass vessels due to corrosion?
Name one metal each occurring as :
An oxide
Bauxite is mixed with cryolite so as to
(i) reduce its melting point
(ii) increase its electrical conductivity
(iii) molten cryolite acts as a solvent
(iv) increase its melting point
During electrolytic refining of zinc, it gets
Reaction between X and Y, forms compound Z. X loses electron and Y gains electron. Which of the following properties is not shown by Z?
Answer the following question with reference to the electrorefining of copper:
What do you observe at the cathode?
