Advertisements
Advertisements
Question
How does applying pressure (or compression) help in the liquefaction of a gas ?
Solution
We can liquefy gases either by applying pressure (compression). There is a lot of space between the particles of a gas as they have very high kinetic energy and they keep moving randomly. We reduce the spaces between the particles of a gas by compressing it. If enough pressure is applied, the particles of gases start attracting each other. This force brings about a change in the state of matter from gas to liquid.
APPEARS IN
RELATED QUESTIONS
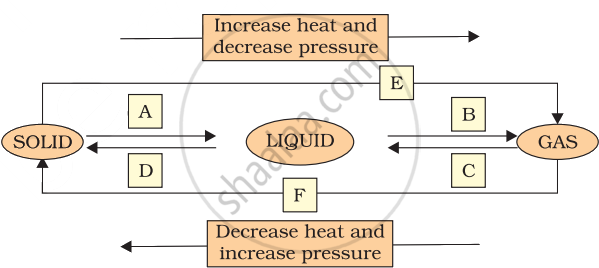
Name A, B, C, D, E and F in the following diagram showing change in its state.
Ripening of fruits is a desirable change.
How do you say that the process of digestion is a chemical change ?
Convert the temperature of 373° C to the Kelvin scale.
Fill in the following blank with suitable words :
Gases can be liquefied by applying ....................... and lowering .....................
Define the following terms: Diffusion
Differentiate between: Gas and vapour
Give two examples of the following:
Substances which sublime.
Explain the term freezing
Write the properties of this substance.
Water
