Advertisements
Advertisements
Questions
How does one explain the emission of electrons from a photosensitive surface with the help of Einstein's photoelectric equation?
Using photon picture of light, show how Einstein’s photoelectric equation can be established.
Solution
n photoelectric effect, an electron absorbs a quantum of energy (hν) of radiation. If this quantum of energy exceeds the minimum energy needed for the electron to escape from the metal surface (work function ϕ0), the electron is emitted with maximum kinetic energy
Kmax = hν - ϕ0
RELATED QUESTIONS
An isolated metal sphere is heated to a high temperature. Will it become positively charged due to thermionic emission?
The anode of a thermionic diode is connected to the negative terminal of a battery and the cathode to its positive terminal.
Define the term "Threshold frequency", in the context of photoelectric emission.
Answer the following question.
Define the term "Threshold frequency", in the context of photoelectric emission.
The wavelength λe of an electron and λp of a photon of same energy E are related by
The wave associated with a moving particle of mass 3 × 10–6 g has the same wavelength as an electron moving with a velocity 6 × 106 ms–1. The velocity of the particle is
Consider Figure for photoemission.
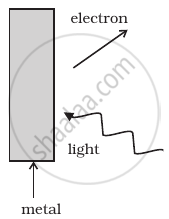
How would you reconcile with momentum conservation? Note light (photons) have momentum in a different direction than the emitted electrons.
Give an example each of a metal from which photoelectric emission takes place when irradiated by
- UV light
- visible light.
Name the factors on which photoelectric emission from a surface depends.
The work function of a metal is 2.31 eV. Photoelectric emission occurs when the light of frequency 6.4 × 1014 Hz is incident on the metal surface. Calculate
- the energy of the incident radiation,
- the maximum kinetic energy of the emitted electron and
- the stopping potential of the surface.
