Advertisements
Advertisements
Question
An isolated metal sphere is heated to a high temperature. Will it become positively charged due to thermionic emission?
Solution
Yes, it will become positively charged due to thermionic emission. When the metal sphere is heated, average kinetic energy of the electrons will increases, due to which, the free electrons of the metal sphere will be able to escape. As a result, they will leave a positive charge on the isolated metal sphere.
APPEARS IN
RELATED QUESTIONS
Find the maximum frequency of X-rays produced by 30 kV electrons.
How does one explain the emission of electrons from a photosensitive surface with the help of Einstein's photoelectric equation?
The work function of the following metals is given : Na 2.75 ev, K = 2.3 eV, Mo = 4.17 eV and Ni = 5.15 eV. Which of these metals will not cause photoelectric emission for radiation of wavelength 3300 Å from a laser source placed 1 m away from these metals? What happens if the laser source is brought nearer and placed 50 cm away?
How will the thermionic current vary if the filament current is increased?
A diode value is connected to a battery and a load resistance. The filament is heated, so that a constant current is obtained in the circuit. As the cathode continuously emits electrons, does it become more and more positively charged?
Why does thermionic emission not take place in non-conductors?
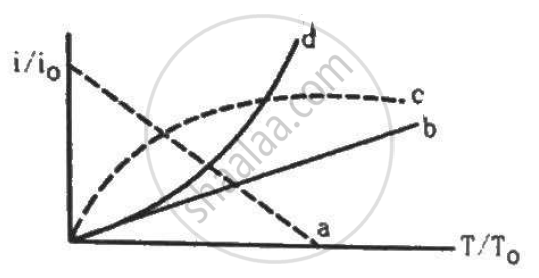
Let i0 be the thermionic current from a metal surface when the absolute temperature of the surface is T0. The temperature is slowly increased and the thermionic current is measured as a function of temperature. Which of the following plots may represent the variation in (i/i0) against (T/T0)?
The anode of a thermionic diode is connected to the negative terminal of a battery and the cathode to its positive terminal.
Define the term "Threshold frequency", in the context of photoelectric emission.
The wavelength λe of an electron and λp of a photon of same energy E are related by
The wave associated with a moving particle of mass 3 × 10–6 g has the same wavelength as an electron moving with a velocity 6 × 106 ms–1. The velocity of the particle is
If a light of wavelength 330 nm is incident on a metal with work function 3.55 eV, the electrons are emitted. Then the wavelength of the wave associated with the emitted electron is (Take h = 6.6 × 10–34 Js)
Emission of electrons by the absorption of heat energy is called ____________ emission.
Why do metals have a large number of free electrons?
What do you mean by electron emission? Explain briefly various methods of electron emission.
In which case is electron emission from a metal not known?
Give an example each of a metal from which photoelectric emission takes place when irradiated by
- UV light
- visible light.
The work function of a metal is 2.31 eV. Photoelectric emission occurs when the light of frequency 6.4 × 1014 Hz is incident on the metal surface. Calculate
- the energy of the incident radiation,
- the maximum kinetic energy of the emitted electron and
- the stopping potential of the surface.
