Advertisements
Advertisements
Question
Would you prefer a material with a high work-function or a low work-function to be used as a cathode in a diode?
Solution
We will prefer a material with low work-function to be used as a cathode in a diode, so that electron emission can occur using a small amount of energy.
APPEARS IN
RELATED QUESTIONS
Find the maximum frequency of X-rays produced by 30 kV electrons.
Find the (a) maximum frequency, and (b) minimum wavelength of X-rays produced by 30 kV electrons.
The work function of caesium metal is 2.14 eV. When light of frequency 6 × 1014 Hz is incident on the metal surface, photoemission of electrons occurs. What is the
(a) maximum kinetic energy of the emitted electrons,
(b) Stopping potential, and
(c) maximum speed of the emitted photoelectrons?
Quarks inside protons and neutrons are thought to carry fractional charges [(+2/3)e; (–1/3)e]. Why do they not show up in Millikan’s oil-drop experiment?
How will the thermionic current vary if the filament current is increased?
An isolated metal sphere is heated to a high temperature. Will it become positively charged due to thermionic emission?
A diode value is connected to a battery and a load resistance. The filament is heated, so that a constant current is obtained in the circuit. As the cathode continuously emits electrons, does it become more and more positively charged?
Why does thermionic emission not take place in non-conductors?
The cathode of a diode valve is replaced by another cathode of double the surface area. Keeping the voltage and temperature conditions the same, will the place current decrease, increase or remain the same?
Let i0 be the thermionic current from a metal surface when the absolute temperature of the surface is T0. The temperature is slowly increased and the thermionic current is measured as a function of temperature. Which of the following plots may represent the variation in (i/i0) against (T/T0)?
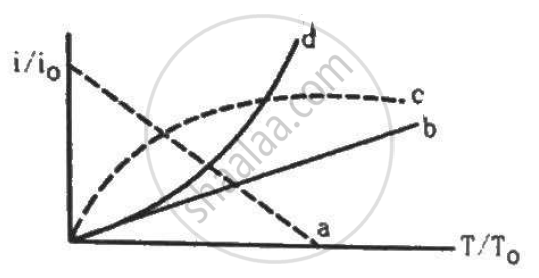
Answer the following question.
Define the term "Threshold frequency", in the context of photoelectric emission.
The wave associated with a moving particle of mass 3 × 10–6 g has the same wavelength as an electron moving with a velocity 6 × 106 ms–1. The velocity of the particle is
If a light of wavelength 330 nm is incident on a metal with work function 3.55 eV, the electrons are emitted. Then the wavelength of the wave associated with the emitted electron is (Take h = 6.6 × 10–34 Js)
Emission of electrons by the absorption of heat energy is called ____________ emission.
Consider Figure for photoemission.
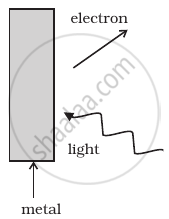
How would you reconcile with momentum conservation? Note light (photons) have momentum in a different direction than the emitted electrons.
Give an example each of a metal from which photoelectric emission takes place when irradiated by
- UV light
- visible light.
