Advertisements
Advertisements
Question
Identify the compounds A, B, C, D, E, and F:
\[\ce{CH3COCH3->[Conc.HNO3][(O)]A}\]
\[\ce{->[SOCl2]B}\]
\[\ce{->[NH3]C}\]
\[\ce{->[LiAlH4]D}\]
\[\ce{->[HNO2]E}\]
\[\ce{->[CH3COCl]F}\]
Solution
Identifying compounds:
\[\begin{array}{cc}\phantom{........................}\ce{O}\\\phantom{........................}||\\\ce{CH3COCH3->[Conc.HNO3][(O)]C\underset{(A)}{H3-C-OH}}\end{array}\]
\[\begin{array}{cc}\phantom{........}\ce{O}\\\phantom{........}||\\\ce{->[SOCl2]C\underset{(B)}{H3-C - Cl}}\end{array}\]
\[\begin{array}{cc}\phantom{......}\ce{O}\\\phantom{......}||\\\ce{->[NH3]\underset{(C)}{CH3-C-NH2}}\end{array}\]
\[\ce{->[LiAlH4]\underset{(D)}{CH3CH2NH2}}\]
\[\ce{->[HNO2]\underset{(E)}{C2H5OH}}\]
\[\begin{array}{cc}\phantom{..............}\ce{O}\\\phantom{..............}||\\\ce{->[CH3COCl]C2H5\underset{(F)}{-O-C-CH3}}\end{array}\]
APPEARS IN
RELATED QUESTIONS
IUPAC name of K4[Fe(CN)6] is
Write the IUPAC name of the given compound:
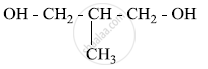
Write down the IUPAC name of the complex [Pt(en)2Cl2]2+. What type of isomerism is shown by this complex?
Write the IUPAC name of the complex [Cr(NH3)4 Cl2]Cl.
Specify the oxidation number of the metal in the following coordination entity:
[Cr(NH3)3Cl3]
Using IUPAC norms write the systematic name of the following:
[Co(NH3)6]Cl3
Using IUPAC norms, write the systematic name of the following:
[Ti(H2O)6]3+
Name the type of isomerism shown by the following compounds:
[CU(NH3)4] [PtCl4] and [Pt(NH3)4] [CuCl4]
Which of the following is paramagnetic?
The complex Hg[Co(CNS)4] is correctly named as ______.
