Advertisements
Advertisements
Question
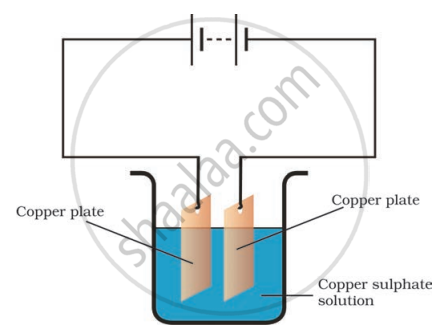
In the circuit given in the figure,
Boojho observed that copper is deposited on the electrode connected to the negative terminal of the battery.
Paheli tried to repeat the same experiment. But she could find only one copper plate. Therefore, she took a carbon rod as negative electrode. Will copper be still deposited on the carbon rod? Explain your answer.
Solution
Yes, Copper from the copper sulphate solution will be deposited on the carbon rod. When electric current is passed through the copper sulphate solution, copper sulphate dissociates into copper and sulphate.
The free copper gets drawn to the electrode connected to the negative terminal of the battery, i.e. carbon rod and gets deposited on it. Thus, Paheli will obtain a coating of copper on carbon rod.
APPEARS IN
RELATED QUESTIONS
If you pass current through copper sulphate solution, copper gets deposited on the plate connected to the ______ terminal of the battery.
The process of depositing a layer of any desired metal on another material by means of electricity is called ______.
Which of the following metals is used in electroplating to make objects appear shining?
The object to be electroplated is taken as ______ electrode.
Observe the following circuits carefully. In which circuit will the bulb glow? Write Yes or No in the blank space provided along each of the circuit given in the figure.
|
(a) |
Piece of coal |
______ |
|
(b) |
Iron nail |
______ |
|
(c) |
Eraser |
______ |
|
(d) |
Steel spoon |
______ |
A chemical reaction happens when electricity passes through various conducting liquids.
Explain the following:
Short circuit.
The terminal which is connected to a positive terminal of a battery is called ______.
Liquids that conduct electricity are the solutions of ______.
Assertion: Parts of cars and bicycles are made iron with chromium coating.
Reason: Chromium does not corrode and resist scratches.




