Advertisements
Advertisements
Question
The object to be electroplated is taken as ______ electrode.
Solution
The object to be electroplated is taken as Negative electrode.
Explanation -
The object to be electroplated is taken as negative electrode or cathode, so that the free ions get deposited on it.
APPEARS IN
RELATED QUESTIONS
If you pass current through copper sulphate solution, copper gets deposited on the plate connected to the ______ terminal of the battery.
The process of depositing a layer of any desired metal on another material by means of electricity is called ______.
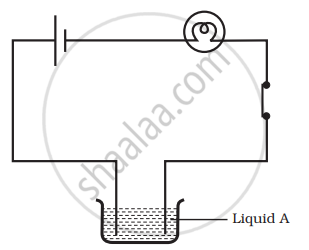
Name three liquids, which when tested in the manner shown in Figure, may cause the magnetic needle to deflect.
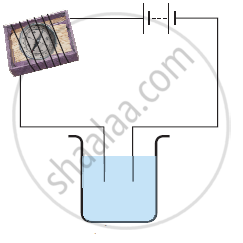
Paheli set up an experiment using liquid A in the beaker as shown in the figure. She observed that the bulb glows. Then, she replaced the liquid A by another liquid B. This time the bulb did not glow. Boojho suggested replacing the bulb by an LED. They observed that the LED glows. Explain.
Why is tin electroplated on iron to make cans used for storing food?
In the circuit given in the figure,
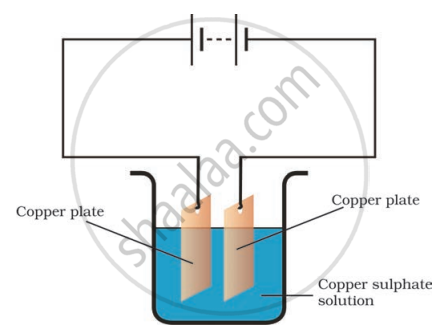
Boojho observed that copper is deposited on the electrode connected to the negative terminal of the battery.
Paheli tried to repeat the same experiment. But she could find only one copper plate. Therefore, she took a carbon rod as negative electrode. Will copper be still deposited on the carbon rod? Explain your answer.
The most common industrial application of chemical effects of electric current is ______.
Liquids that conduct electricity are the solutions of ______.
Assertion: Parts of cars and bicycles are made iron with chromium coating.
Reason: Chromium does not corrode and resist scratches.
What are anodes and cathodes?
