Advertisements
Advertisements
Question
One of the most common applications of chemical effect of electric current is ______.
Solution
One of the most common applications of chemical effect of electric current is Electroplating.
Explanation -
It is the process of depositing a layer of any desired metal on another material by means of electricity.
APPEARS IN
RELATED QUESTIONS
If you pass current through copper sulphate solution, copper gets deposited on the plate connected to the ______ terminal of the battery.
Prepare a list of objects around you that are electroplated.
The process that you saw in the image is used for the purification of copper. A thin plate of pure copper and a thick rod of impure copper are used as electrodes. Copper from impure rod is sought to be transferred to the thin copper plate. Which electrode should be attached to the positive terminal of the battery and why?
Which of the following metals is used in electroplating to make objects appear shining?
Electroplating of ______ is done on objects like water taps and cycle bell to give them a shiny appearance.
Paheli set up an experiment using liquid A in the beaker as shown in the figure. She observed that the bulb glows. Then, she replaced the liquid A by another liquid B. This time the bulb did not glow. Boojho suggested replacing the bulb by an LED. They observed that the LED glows. Explain.
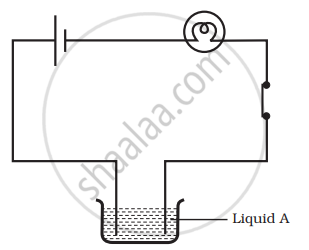
Observe the following circuits carefully. In which circuit will the bulb glow? Write Yes or No in the blank space provided along each of the circuit given in the figure.
|
(a) |
Piece of coal |
______ |
|
(b) |
Iron nail |
______ |
|
(c) |
Eraser |
______ |
|
(d) |
Steel spoon |
______ |
A chemical reaction happens when electricity passes through various conducting liquids.
The terminal which is connected to a positive terminal of a battery is called ______.
Assertion: Parts of cars and bicycles are made iron with chromium coating.
Reason: Chromium does not corrode and resist scratches.




