Advertisements
Advertisements
Question
In the redox reaction, \[\ce{2MnO^-_4 + SO^{2-}_3 + H2O -> 2MnO^{2-}_4 + SO^{2-}_4 + 2H^+}\], the reductant is:
Options
\[\ce{MnO^-_4}\]
\[\ce{SO^{2-}_3}\]
\[\ce{H2O}\]
\[\ce{H^+}\]
MCQ
Solution
\[\ce{SO^{2-}_3}\]
Explanation:
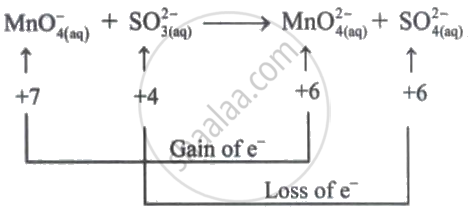
The oxidation number of 'S' is increased from +4 to +6. Therefore, \[\ce{SO^{2-}_3}\] is the reducing agent/reductant.
shaalaa.com
Redox Reactions
Is there an error in this question or solution?
