Advertisements
Advertisements
Question
In the SF4 molecule, there are:
Options
4 lone pairs and one bond pair and shape is tetrahedral
4 bond pairs and one lone pair and shape is see-saw
2 lone pairs and four bond pair and shape is linear
1 lona pairs and 6 bond pair and shape is octahedral
MCQ
Solution
4 bond pairs and one lone pair and shape is see-saw
Explanation:
No. of bond pair: 4
No. of lone pair: 1
Shape - See saw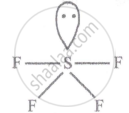
SF4 (sp3d hyb.)
shaalaa.com
Close Packed Structures of Solids
Is there an error in this question or solution?
