Advertisements
Advertisements
Question
Is the following sugar, D-sugar or L-sugar?
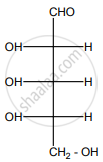
Solution
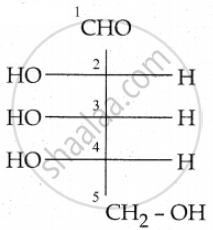
L-sugar
\[\begin{array}{cc}
\phantom{.....}\ce{^1CHO}\\
\phantom{...}|\\
\ce{HO - ^2C - H}\\
\phantom{...}|\\
\phantom{........}\ce{\underset{L-Glyceraldehyde}{^3CH2OH}}
\end{array}\]
Because the H and OH on C4 carbon is in the same configuration as the H and OH on C2
carbon in L-Glyceraldehyde.
APPEARS IN
RELATED QUESTIONS
Write a chemical reaction to convert glucose into glucose cyanohydrin.
Which among the following compounds is obtained when glucose reacts with hydrogen cyanide?
4-O-(α-D-Glucopyranosyl)-D-glucopyranose is ____________.
Which of the following monosaccharide is a ketohexose?
Which one of the following carbohydrates is insoluble in water?
Glucose and gluconic acid treated with dilute nitric acid forms saccharic acid. What does this indicate?
What is the molecular formula of glyceraldehyde?
Which one given below is a non-reducing sugar?
\[\ce{Glucose ->[(HCN)] Product ->[(hydrolysis)] Product ->[(HI + Heat)] A}\], the compound A is:
Glucose is an aldose. Which one of the following reactions is not expected with glucose?
