Advertisements
Advertisements
Question
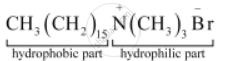
Label the hydrophilic and hydrophobic parts in the following compounds.
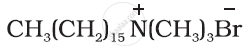
Solution
APPEARS IN
RELATED QUESTIONS
Explain the following term with a suitable example:
Cationic detergents
Explain the following terms with suitable examples - Anionic detergents
Label the hydrophilic and hydrophobic parts in the following compounds.
CH3(CH2)10CH2OSO3 –Na+
Label the hydrophilic and hydrophobic parts in the following compounds.
`CH_3(CH_2)_16COO(CH_2CH_2O)_nCH_2CH_2OH`
Define disinfectants with a Suitable example?
Define the following term with a suitable example:
Cationic detergents
What type of detergent are used in toothpastes?
Which of the following is an example of liquid dishwashing detergent?
Polyethyleneglycols are used in the preparation of which type of detergents?
Which of the following are anionic detergents?
(i) Sodium salts of sulphonated long-chain alcohol.
(ii) Ester of stearic acid and polyethylene glycol.
(iii) Quarternary ammonium salt of amine with acetate ion.
(iv) Sodium salts of sulphonated long-chain hydrocarbons.
Synthetic detergents have advantage over usual soaps as far as cleansing power is concerned. But use of synthetic detergents over a long time creates environmental pollution. How can the pollution caused by synthetic detergents be minimised? Classify the detergents according to their chemical nature.
Explain the following term with suitable example.
Cationic detergents
Define the following:
Cationic detergents
Explain the following term with suitable examples.
cationic detergents
Explain the following term with a suitable example.
cationic detergents
Explain the following term with suitable examples.
Cationic detergents
Explain the following term with suitable example.
Cationic detergents
Explain the following term with suitable example:
cationic detergents
