Advertisements
Advertisements
Question
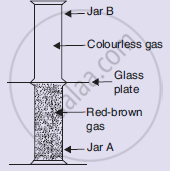
Look at the diagram on the right side. Jar A contains a red-brown gas whereas jar B contains a colourless gas. The two gas jars are separated by a galas plate placed between them
- What will happen when the glass plate between the two jars is pulled away?
- What name is given to the phenomenon which takes place?
- Name the brown gas which could be in jar A.
- Which is the colourless gas most likely to be present in jar B?
- Name one coloured solid and one colourless liquid which can show the same phenomenon.
Solution
- When the glass plate between the two jars is pulled away then the red brown gas will diffuse from jar A into colourless jar B and vice versa, due to which the red brown colour will spread into jar B.
- The phenomenon occurring here is diffusion.
- Bromine vapour.
- Air.
- Potassium permanganate (coloured solid) and water (colourless liquid) also show the same phenomenon.
APPEARS IN
RELATED QUESTIONS
Give a reason for the following observation.
We can get the smell of perfume sitting several metres away.
Arrange the following substances in increasing order of forces of attraction between particles -
water, sugar, oxygen
Name the physical state of matter which can be easily compressed.
Fill in the following blank with suitable words :
At room temperature, the forces of attraction between the particles of solid substances are ....................... then those which exist in the gaseous state.
Why do gases have neither a fixed shape nor a fixed volume?
Give one example of diffusion of gases in a liquid.
Explain why, when a bottle of perfume is opened in a room, we can smell it even from a considerable distance.
Name two gases of air which dissolve in water by diffusion. What is the importance of this process in nature?
A substance has neither a fixed shape nor a fixed volume. State whether it is a solid, a liquid or a gas.
2 litre He gas at 2 atm and 300 K is inserted into a 4 litre rigid container containing N2 at 600 K and 4 atm. Finally mixture is maintained at 600 K temperature. The final pressure of gaseous mixture would be ______ torr.
