Advertisements
Advertisements
Question
Explain why, when a bottle of perfume is opened in a room, we can smell it even from a considerable distance.
Solution
When a bottle of perfume is opened in a room, the particles of perfume, start diffusing into the air surrounding the bottle. These particles of perfume keep moving towards a region of lower concentration by the virtue of their kinetic energy. Thus, when a bottle of perfume is opened, we can smell perfume even from a distance.
APPEARS IN
RELATED QUESTIONS
The mass per unit volume of a substance is called density.
(density = mass/volume).
Arrange the following in order of increasing density-
air, exhaust from chimneys, honey, water, chalk, cotton and iron.
Which of the two diffuses faster : a liquid or a gas?
If the fish is being fried in a neighbouring home, we can smell it sitting in our own home. Name the process which brings this smell to us.
Name the three states of matter. Give one example of each.
Give two reasons to justify that Water is a liquid at room temperature.
Why do gases diffuse very fast?
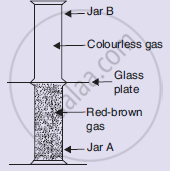
Look at the diagram on the right side. Jar A contains a red-brown gas whereas jar B contains a colourless gas. The two gas jars are separated by a galas plate placed between them
- What will happen when the glass plate between the two jars is pulled away?
- What name is given to the phenomenon which takes place?
- Name the brown gas which could be in jar A.
- Which is the colourless gas most likely to be present in jar B?
- Name one coloured solid and one colourless liquid which can show the same phenomenon.
The substance X normally exists in a physical state which can flow easily but does not fill its vessel completely. It also turns anhydrous copper sulphate blue. When substance X is cooled excessively, it changes into a substance Y which has a fixed shape as well as a fixed volume. If, however, the substance X is heated strongly, it changes into a substance Z which has neither a fixed shape nor a fixed volume.
- Name the substances (i) X (ii) Y and (iii) Z.
- What is the process of conversion of X into Y known as ?
- At which temperature X gets converted into Y ?
- What is the process of conversion of X into Z known as ?
- At which temperature X gets converted into Z ?
Comment upon the following:-
fluidity
A gaseous mixture of He and O2 is found to have a density of 0.518 gL-1 at 25° C and 720 torr. The mass percent of helium in this mixture is ______.
