Advertisements
Advertisements
Question
If the fish is being fried in a neighbouring home, we can smell it sitting in our own home. Name the process which brings this smell to us.
Solution
The vapours of the fried fish travel from one place to another through the process of diffusion.
APPEARS IN
RELATED QUESTIONS
Give reasons:
A gas fills completely the vessel in which it is kept.
Give a reason for the following observation.
We can get the smell of perfume sitting several metres away.
State two characteristic properties of a gas.
A girl is cooking some food in the kitchen. The smell of food being cooked soon reaches her brother's room. Explain how the smell could have reached her brother's room.
Give one example of diffusion of gases in a liquid.
Give reason for the following observation :
The smell of hot sizzling food reaches us even from a considerable distance but to get the smell from cold food, we have to go close to it.
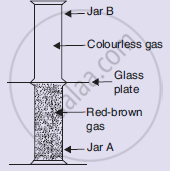
When a gas jar containing colourless air is kept upside down over a gas jar full of brown-coloured bromine vapour, then after some time, the brown colour of bromine vapour spreads into the upper gas jar making both the gas jars appear brown in colour. Which of the following conclusion obtained from these observations is incorrect?
Look at the diagram on the right side. Jar A contains a red-brown gas whereas jar B contains a colourless gas. The two gas jars are separated by a galas plate placed between them
- What will happen when the glass plate between the two jars is pulled away?
- What name is given to the phenomenon which takes place?
- Name the brown gas which could be in jar A.
- Which is the colourless gas most likely to be present in jar B?
- Name one coloured solid and one colourless liquid which can show the same phenomenon.
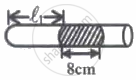 |
|
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)
Two gases A and B having molecular weights 60 and 45 respectively are enclosed in a vessel. The wt. of A is 0.50 g and that of B is 0.2 g. The total pressure of the mixture is 750 mm. The partial pressure of the gases B is ______ mm Hg.