Advertisements
Advertisements
Question
Name the reagents used in the following reactions:
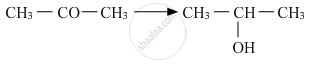
Solution
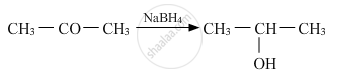
Name of the reagent: Sodium borohydride (NaBH4)
APPEARS IN
RELATED QUESTIONS
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{......}\\
\ce{CH3}\phantom{...}
\end{array}\]
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
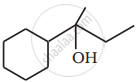
Show how you would synthesise the following alcohol from an appropriate alkene?
Write the structures of A, B and C in the following reactions :
Ketones react with Grignard reagent to produce ____________.
Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
The Wittig reaction is a reaction between a carbonyl compound (aldehyde or ketone only) and a species known as a phosphoniumylide. What is the expected final product in the Wittig reaction?
The best reagent to convert pent - 3 - en - 2 - ol into pent - 3 - en - 2 one is ______.
Ceric aminonium nitrate and CHCl3/alc. KOH are used for the identification of functional groups present in ______ and ______ respectively.
