Advertisements
Advertisements
Question
Observe the following cell and answer the questions that follow:
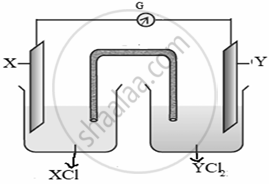
- Represent the cells shown in the figure.
- Name the carriers of the current in the salt bridge.
- Write the reaction taking place at the anode.
Chemical Equations/Structures
Short Answer
Solution
- \[\ce{Y(s)|Y^{2+}(aq) || X+(aq)| X(s)}\]
- Ions are carriers of current in the salt bridge.
- \[\ce{Y(s) -> Y^{2+}(aq) + 2e−}\]
shaalaa.com
Is there an error in this question or solution?
