Advertisements
Advertisements
Question
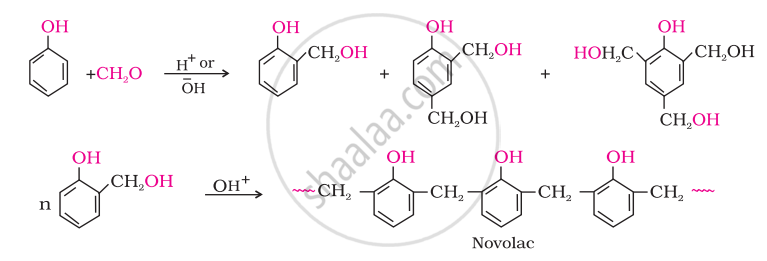
Phenol and formaldehyde undergo condensation to give a polymar (A) which on heating with formaldehyde gives a thermosetting polymer (B). Name the polymers. Write the reactions involved in the formation of (A). What is the structural difference between two polymers?
Solution
Phenol–formaldehyde polymers are the oldest synthetic polymers. These are obtained by the condensation reaction of phenol with formaldehyde in the presence of either an acid or a base catalyst. The reaction starts with the initial formation of o-and/or p-hydroxymethyl phenol derivatives, which further react with phenol to form compounds having rings joined to each other through –CH2 groups. The initial product could be a linear product – Novolac
Novolac on heating with formaldehyde undergoes cross-linking to form an infusible solid mass called bakelite. It is used for making combs, phonograph records, electrical switches and handles of various utensils.
'A' is Novolac and 'B' is Bakelite.

APPEARS IN
RELATED QUESTIONS
Write the structures of the monomers used for getting the following polymers
Melamine – formaldehyde polymer
Choose the correct option from the given alternatives.
Which of the following is made up of polyamides?
Identify the polymer obtained by polymerization of n moles of acrylonitrile.
Which among the following polymers can NOT be remoulded?
Which of the following pair of compounds is used as monomers for bakelite?
Identify addition polymer from the following.
The commercial name of polyacrylonitrile is ______.
Match the following pairs:
| Polymer | Monomer | ||
| (i) | Teflon | (a) | CH2 = CH2 |
| (ii) | PVC | (b) | CF2 = CF2 |
| (iii) | Natural rubber | (c) | Isoprene |
| (iv) | Polythene | (d) | H2C=CHCl |
Write the structure and name of monomer of Nylon-6.
Name and draw structure of the repeating unit in natural rubber.
