Advertisements
Advertisements
Question
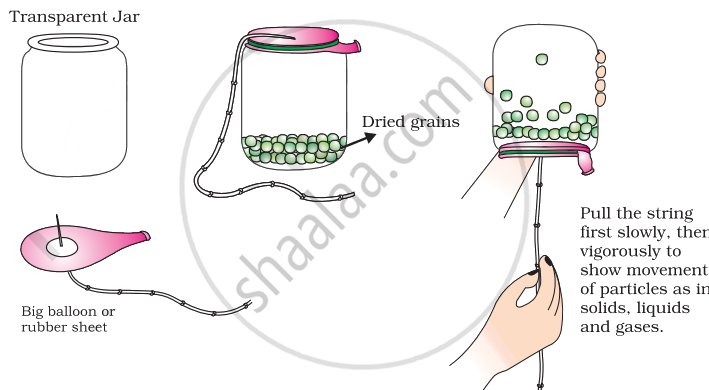
Prepare a model to demonstrate movement of particles in solids, liquids and gases.
For making this model you will need
- A transparent jar
- A big rubber balloon or piece of stretchable rubber sheet
- A string
- Few chickpeas or black gram or dry green peas.
How to make?
- Put the seeds in the jar.
- Sew the string to the centre of the rubber sheet and put some tape to keep it tied securely.
- Stretch and tie the rubber sheet on the mouth of the jar.
- Your model is ready. Now run your fingers up and down the string by first tugging at it slowly and then rapidly.
Solution
Materials Required:
- A transparent jar
- A big rubber balloon or piece of stretchable rubber sheet
- A string
- Few chickpeas, black gram, or dry green peas
Steps to Make the Model:
-
Prepare the Jar:
- Take a transparent jar and put the dried seeds (chickpeas, black gram, or dry green peas) into the jar. These seeds will represent the particles.
-
Attach the Rubber Sheet:
- Sew a string to the centre of the rubber balloon or rubber sheet.
- Secure the string tightly using some tape to ensure it stays attached.
- Stretch the rubber sheet over the mouth of the jar and tie it securely. This setup allows the rubber sheet to simulate the movement when the string is pulled.
-
Demonstrating Particle Movement:
- Your model is now ready.
- Gently pull the string attached to the rubber sheet and observe the movement of the seeds inside the jar.
- Start by tugging the string slowly to demonstrate the movement of particles in solids, where they move slightly.
- Then, pull the string more vigorously to demonstrate the movement of particles in liquids and gases, where they move more freely and rapidly.
This model effectively shows how particles in solids, liquids, and gases behave differently depending on the energy (represented by the speed of string pulling) applied to them.
RELATED QUESTIONS
Convert the following temperature to Celsius scale:
300 K
Is there any change in temperature during the change of phase?
Name one property which is shown by ammonium chloride but not by sodium chloride.
Which contains more heat, 1 kg of water at 100° C or 1 kg of steam at 100° C ? Give reason for your answer.
Define 'boiling point' of a substance ? What is the boiling point of water ?
Which of the following energy is absorbed during the change of state of a substance ?
Match the following:
| Column A | Column B |
| (a) Evaporation of water | (i) Non-periodic change |
| (b) Milk turning sour | (ii) Periodic change |
| (c) Earthquake | (iii) Chemical change |
| (d) Change of seasons | (iv) Physical change |
| (e) Glowing of bulb | (v) Reversible change |
Define: cohesive force
Define the following terms: Condensation
Name the phenomenon which causes the following changes:
Conversion of ice into water
