Advertisements
Advertisements
Question
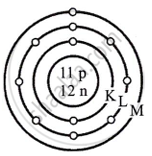
State the atomic number and mass number of \[\ce{^23_11Na}\] and draw its atomic model.
Diagram
Short Answer
Solution
Atomic number Z = 11
Mass number A = 23
A = Protons + Neutrons
23 = 11 + Number of neutrons
Number of neutrons = 23 - 11 = 12
Atomic model of sodium Na
11 = 2, 8, 1
shaalaa.com
Is there an error in this question or solution?
APPEARS IN
RELATED QUESTIONS
Electromagnetic radiation of wavelength 242 nm is just sufficient to ionise the sodium atom. Calculate the ionisation energy of sodium in kJ mol–1.
The diameter of zinc atom is 2.6 Å. Calculate
- radius of zinc atom in pm and
- number of atoms present in a length of 1.6 cm if the zinc atoms are arranged side by side length-wise.
What is nucleus of an atom? Compare its size with that of the atom. Name its constituents. How is the number of these constituents determined by the atomic number and mass number of the atom?
