Advertisements
Advertisements
Question
State your observation for the following cases
(i) Ammonia gas is burnt in an atomosphere of oxygen in the absence of a catalyst
(ii) Glass rod dipped in ammonium hydroxide is brought near the mouth of the concentrated hydrochloric acid bottle
Solution
(i) Ammonia burns with a yellowish flame. It produces water vapour and nitrogen.
(ii) When ammonium hydroxide is brought near the mouth of concentrated hydrochloric acid, it produces dense white fumes of ammonium chloride.
APPEARS IN
RELATED QUESTIONS
What property of ammonia is demonstrated by the fountain experiment?
When an ammonium salt is warmed with a sodium hydroxide solution, a gas is evolved. State three ways in which you can identify this gas.
What is liquor ammonia fortis?
Choose the correct word or phrase from the bracket to complete the following sentences.
Heating solution of ammonium chloride with sodium nitrite produces ______
The diagram shows a simple arrangement of the fountain experiment:
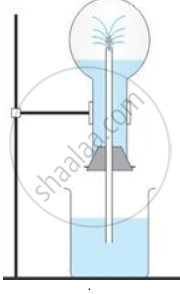
(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
What do you observe when Ammonia in excess is mixed with chlorine.
Of the two gases, ammonia and hydrogen chloride, which is denser? Name the method of collection of this gas.
Identify the terms for the following:
The experiment which demonstrates high solubility of ammonia gas.
