Advertisements
Advertisements
Question
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 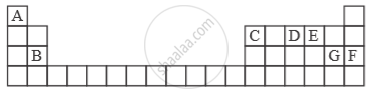
Which non-metallic element has the valency of 2?
Solution
Non-metallic element valency of 2 -‘E’.
APPEARS IN
RELATED QUESTIONS
Metallic character and non-metallic character are periodic properties discuss ?
Give the trend in metallic character:
(i) across the period left to right
Explain the following:
Group 17 elements are strong non-metals, while group 1 elements are strong metals.
Explain the following:
Metallic character of elements decreases from left to right in a period while it increases in moving down a group.
A metal M forms as oxide having the formula M2O3. It belongs to third period. Write the atomic number and valency of the metal.
For the main group of the periodic table, the metallic properties of the elements vary approximately with their position as shown in the table.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| H | He | ||||||
| A | B | ||||||
| C | D |
Will the most metallic element be found at A,B,C or D ?
The tendency of an element to form anion is the ______ character of that element.
3, 1, 2 electrons are in valence shells of X, Y, Z elements. From this information, state the group in which they belong and write their valencies.
An element has atomic number 17. To which group, the period does it belong? It is metal or non-metal?
The given table shows elements with the same number of electrons in its valence shell.
| Elements | A | B | C |
| m.p. | 63.0 | 180.0 | 97.0 |
Arrange them in order of increasing metallic character.
