Advertisements
Advertisements
Question
Substances which conduct electricity in the solid state are generally ______.
Solution
Substances which conduct electricity in the solid state are generally metals.
APPEARS IN
RELATED QUESTIONS
Name the following:
An alloy of lead and tin that is used in electrical circuits.
A solution of HCl gas in water conducts electricity because ______, but a solution of HCl gas in toluene does not conduct an electric current because ______.
Define the following term:
Non-electrolyte
Name a salt which is a weak electrolyte.
What is meant by the term 'electrolyte'?
Write down the word or phrase from the given options that will correctly fill in the blanks in the following sentence:
Pure water consists entirely of ________
Fill in the blank from the choices given below :
In covalent compounds, the bond is formed due to the ______ of electrons.
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
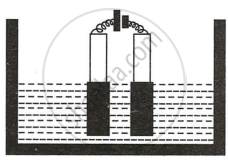
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Name the metallic ions that should be present in the electrolyte when an article made copper is to be electroplated with silver
Select the ion, that would get selectively discharge from the aqueous mixture of the ions listed below :
Pb2+, Ag+, Cu+
