Advertisements
Advertisements
Question
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
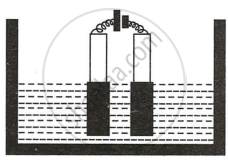
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Solution
- The electrode on the left (anode) side donates electrons and hence is the oxidizing electrode.
- \[\ce{Cu - 2e- -> Cu^{2+}}\]
- The anode size steadily lowers while the cathode size gradually increases. However, the colour of the copper sulphate solution does not change.
APPEARS IN
RELATED QUESTIONS
State the factors that influence the preferential discharge of ions at the electrodes.
Explain the term : Anode mud
Explain, why during the electrolysis of copper sulphate using copper electrodes, the colour of solution does not fade?
How is electrolytic dissociation different from thermal dissociation?
What are the particles present in a compound which is a non- electrolyte?
Explain, why copper though a good conductor of electricity is, a non- electrolyte.
Fill in the blank.
As we descend the electrochemical series containing cations, the tendency of the cations to get ________ at the cathode increases.
Select the correct answer from the choices a,b,c and d which are given. Write only the letter corresponding to the correct answer.
A compound which liberates reddish brown gas around the anode during the electrolysis in its molten state is:______________
Fill in the blank from the choices given below :
Electro covalent compounds have a _____ boiling point
When fused lead bromide is electrolyzed we observe
