Advertisements
Advertisements
Question
State the factors that influence the preferential discharge of ions at the electrodes.
Solution
The following are the factors that influence the preferential discharge of ions at the electrode:
i) Position of the metallic ion in electrochemical series : If all the factors remain the same, an ion placed lower in the electrochemical series gets preferentially discharged at the respective electrode in comparison to all those ios, which are placed above it in the series.
ii) Concentration of ions in the electrolyte : Higher the
concentration of negative ion in the electrolytic solution, greater is its probability of being discharged at the anode.
iii) Nature of the electrode : If the electrode used is inert i,e made of less reactive material such as graphite, platinum etc, the electrode does not play any role in deciding the preferential discharge of an ion at it.
If the electrode used is active i,e made of active material such as Cu, Ag, Ni etc it takes part in the electrode reaction and plays an important role in deciding the ions which will preferentially be discharged. In such a case, anions migrate to the anode but do not get discharged ,Instead the active anode itself loses electrons and form ions.
APPEARS IN
RELATED QUESTIONS
Name the following:
An alloy of lead and tin that is used in electrical circuits.
Name the gas that is produced in each of the given case :
At the anode during the electrolysis of acidified water.
The following Question refer to the electrolysis of copper sulphate solution with copper electrodes:
(a) Compare the change in mass of the anode
(b) What is seen to happen to the colour of the copper sulphate solution if platinum electrodes are used? Explain the observation.
(c) What is the practical application of the electrolysis of copper sulphate solution? Briefly, describe one such application.
Study the diagram given alongside and answer the questions that follow :
(i) Give the names of the electrode A and B.
(ii) Which electrode is oxidizing electrode?
Here is an electrode reaction:
\[\ce{Cu -> Cu^{2+} + 2e^-}\]
At which electrode (anode or cathode) would such a reaction take place? Is this an example of oxidation?
Select the correct answer from the choices a,b,c and d which are given. Write only the letter corresponding to the correct answer.
A compound which liberates reddish brown gas around the anode during the electrolysis in its molten state is:______________
Give reasons as to why - the electrolysis of acidulated water is considered to be an example of catalysis.
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
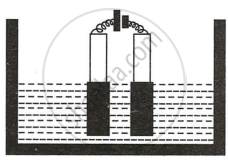
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
M is a metal above hydrogen in the activity series and its oxide has the formula M2O. The oxide when dissolved in water forms the corresponding hydroxide which is a good conductor of electricity. In the above context answer the following:
- What kind of combination exists between M and O?
- How many electrons are there in the outermost shell of M?
- Name the group to which M belongs.
- State the reaction taking place at the cathode.
- Name the product at the anode.
State the observation at the anode and at the cathode during the electrolysis of :
Copper sulphate solution using copper electrodes.
