Advertisements
Advertisements
Question
The \[\ce{C - O - H}\] bond angle in alcohol is ______.
Options
slightly greater than 109°28'
slightly less than 109°28'
slightly greater than 120°
slightly less than 120°
MCQ
Fill in the Blanks
Solution
The \[\ce{C - O - H}\] bond angle in alcohol is slightly less than 109°28'.
Explanation:
\[\ce{C - O - H}\] bond angle in alcohol is slightly less than slightly less than 109°28'.
The oxygen atom is sp3 hybridised, but the resulting bond angle \[\ce{C - O - H}\] is marginally smaller than the tetrahedral angle due to the mutual repulsion of two lone pairs of electrons on it.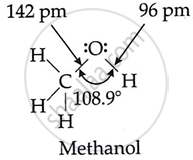
shaalaa.com
Is there an error in this question or solution?
