Advertisements
Advertisements
Question
The electric current in an X-ray tube (from the target to the filament) operating at 40 kV is 10 mA. Assume that on an average, 1% of the total kinetic energy of the electron hitting hte target are converted into X-rays.
(a) What is the total power emitted as X-rays and (b) how much heat is produced in the target every second?
Solution
Given :
V = 30 KV
i = 10mA
1% of TKE (total kinetic energy) = X-ray
i = ne
or `n = (10^-2)/(1.6 xx 10^-19)`
= `0.625 xx 10^17` (number of electrons)
KE of one electron = eV
= `1.6 xx 10^-19 xx 40 xx 10^3`
= `6.4 xx 10^-15 "J"`
`"T"_("KE") = 0.625 xx 6.4 xx 10^17 xx 10^-15`
= `4 xx 10^2 "J"`
(b) Heat produced in target per second = 400 − 4 = 396 J
APPEARS IN
RELATED QUESTIONS
What role dose infra-red radiation play in physical therapy?
What physical quantity is the same for X-rays of wavelength 10−10 m, red light of wavelength 6800 Å and radiowaves of wavelength 500 m?
A radio can tune in to any station in the 7.5 MHz to 12 MHz band. What is the corresponding wavelength band?
Arrange the following radiations in the order of their increasing wavelength:
X-rays, infrared rays, ratio waves, gamma ray and microwaves.
What is the range of the wavelength of the following electromagnetic waves?
(a) Micro waves .
Name the region beyond the red end of the spectrum.
Name two electromagnetic waves of frequency smaller than that of violet light. State one use of each.
If the potential difference applied to the tube is doubled and the separation between the filament and the target is also doubled, the cutoff wavelength
If the current in the circuit for heating the filament is increased, the cutoff wavelength
Moseley's Law for characteristic X-ray is √v = a(Z − b). Here,
The X-ray coming from a Coolidge tube has a cutoff wavelength of 80 pm. Find the kinetic energy of the electrons hitting the target.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Find the maximum potential difference which may be applied across an X-ray tube with tungsten target without emitting any characteristic K or L X-ray. The energy levels of the tungsten atom with an electron knocked out are as follows.
| Cell containing vacancy | K | L | M |
| Energy in keV | 69.5 | 11.3 | 2.3 |
Name the scientist who discovered Visible light
Define the term "Intensity" in the photon picture of electromagnetic radiation.
Answer briefly.
Give two uses of ultraviolet rays.
One requires 11eV of energy to dissociate a carbon monoxide molecule into carbon and oxygen atoms. The minimum frequency of the appropriate electromagnetic radiation to achieve the dissociation lies in ______.
Assertion (A): Ultraviolet radiations is scattered more as compared to the microwave radiations.
Reason (R): Wavelength of ultraviolet radiation is more than the wavelength of microwave radiation.
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.
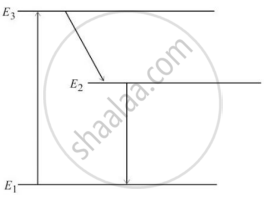
- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
