Advertisements
Advertisements
Question
The reaction of toluene with chlorine in the presence of iron and in the absence of light yields ______.
Options
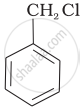
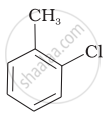
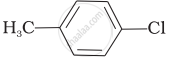
Mixture of
 and
and 
Solution
Mixture of  and
and 
Explanation:
Both ortho- and para- compounds are formed in different yields as depicted below:

('X' represents Cl in this case)
Even though the ortho- product is the major product.
APPEARS IN
RELATED QUESTIONS
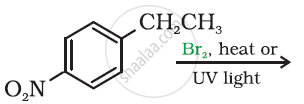
Draw the structure of major monohalo products in the following reaction:
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
(CH3)2CHCH(Cl)CH3
What should be the correct IUPAC name for diethylbromomethane?
Which of the products will be major product in the reaction given below? Explain.
\[\ce{CH3CH = CH2 + HI -> \underset{(A)}{CH3CH2CH2I} + \underset{(B)}{CH3CHICH3}}\]
Why can aryl halides not be prepared by reaction of phenol with \[\ce{HCl}\] in the presence of \[\ce{ZnCl2}\]?
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
