Advertisements
Advertisements
Question
What should be the correct IUPAC name for diethylbromomethane?
Options
1-Bromo-1,1-diethylmethane
3-Bromopentane
1-Bromo-1-ethylpropane
1-Bromopentane
Solution
3-Bromopentane
Explanation:
Diethylbromomethane is
\[\begin{array}{cc}
\phantom{}\ce{Br}\phantom{..}\\
\phantom{}|\phantom{...}\\
\phantom{}\ce{H3\overset{5}{C}- H2\overset{4}{C} - \overset{3}{C}H - \overset{2}{C}H2 - \overset{1}{C}H3}
\end{array}\]
Its IUPAC name is 3-bromopentane.
APPEARS IN
RELATED QUESTIONS
Draw the structure of major monohalo products in the following reaction:
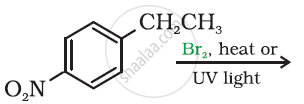
Draw the structure of major monohalo products in the following reaction:
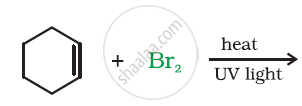
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
(CH3)2CHCH(Cl)CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH(CH3)CH(Br)CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
o-Br-C6H4CH(CH3)CH2CH3
In which of the following molecules carbon atom marked with asterisk (*) is asymmetric?
(a) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(b) \[\begin{array}{cc}
\ce{D}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(c) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{OH}\phantom{..}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
(d) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{H}\phantom{...}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
Ethylidene chloride is a/an ______.
Haloalkanes contain halogen atom (s) attached to the sp3 hybridised carbon atom of an alkyl group. Identify haloalkane from the following compounds.
(i) 2-Bromopentane
(ii) Vinyl chloride (chloroethene)
(iii) 2-chloroacetophenone
(iv) Trichloromethane
Match the items of Column I and Column II.
| Column I | Column II | |
| (i) | SN1 reaction | (a) vic-dibromides |
| (ii) | Chemicals in fire extinguisher | (b) gem-dihalides |
| (iii) | Bromination of alkenes | (c) Racemisation |
| (iv) | Alkylidene halides | (d) Saytzeff rule |
| (v) | Elimination of HX from alkylhalide | (e) Chlorobromocarbons |
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl, or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
